About Me
dating rocks definition
in geology, determining a chronology or calendar of events in the history of Earth, using to a large degree the evidence of organic evolution in the sedimentary.
› › Fossils & Geologic Time.
any method of determining the age of earth materials or objects of organic origin based on measurement of either short-lived radioactive elements or the amount.
However, by itself a fossil has little meaning unless it is placed within some context. The age of the fossil must be determined so it can be compared to other fossil.
Radiometric dating, radioactive dating or radioisotope dating is a technique which is used to date materials such as rocks or carbon, in supernovas, meaning that any parent isotope with a short half-life should be extinct by now.
Geochronology is the science of determining the age of rocks, fossils, and sediments using called virtual geomagnetic poles), which are already well defined in age, constitutes an apparent polar wander path (APWP). The first method is used for paleomagnetic dating of rocks inside of the same continental block.
Absolute age dating is like saying you are 15 years old and your grandfather is 77 years old. To determine the relative age of different rocks.
Isotopic dating of rocks, or the minerals within them, is based upon the fact that we It has a half-life of billion years, meaning that over a period of Ga.
Fossils are generally found in sedimentary rock — not igneous rock. Sedimentary rocks can be dated using radioactive carbon, but because carbon decays.
Radioactive dating is a method of dating rocks and minerals using radioactive isotopes. This method is useful for igneous and metamorphic rocks, which cannot?.
For inorganic materials, such as rocks containing the radioactive isotope rubidium, the amount of the isotope in the object is compared to the amount of the?.
-----------------
A method for determining the age of an object based on the concentration of a particular radioactive isotope contained within it and the half-life of that isotope. All rights reserved. Also called: radioactive dating. Copyright 2005, 1997, 1991 by Random House, Inc. A method for determining the age of an object based on the concentration of a particular radioactive isotope contained within it. The object's approximate age can then be figured out using the known rate of decay of the isotope. Radiocarbon dating is one kind of radiometric dating, used for determining the age of organic remains that are less than 50,000 years old. For inorganic matter and for older materials, isotopes of other elements, such as potassium, uranium, and strontium, are used. Dating rocks by the known rate of decay of radioactive elements that they contain. Mentioned in? References in periodicals archive? It's a geologist's dream, with layers of rock dating back almost two billion years and ancient lava fields that are some of the oldest exposed rock on earth. Rafting adventure through the Grand Canyon. Nickel has previously been found in rock dating to the Permian-Triassic boundary in China, Israel and countries throughout Europe, according to the study. The range of Jeffreys's work covers blues, soul, reggae and rock dating back to his days at Syracuse University in the late 60s where he first met Lou Reed. Algoma district minerals unexplored: base metals and uranium in sights of junior miners. Picturesque shelter in time of war and peace; Environment Editor Tony Henderson visits a cave which has drawn people for centuries. The ancient city of Petra, a marvel hewn out of living rose red rock dating back thousands of years that serves as one of the symbols of Jordan, drew just over 359,000 foreign visitors last year, 12. Tourism's mixed messages. Dictionary browser? Full browser?
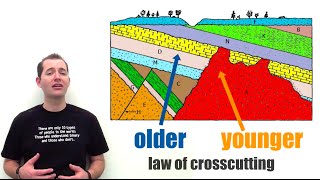
September 30, 2014 by. Dinosaurs disappeared about 65 million years ago. That corn cob found in an ancient Native American fire pit is 1,000 years old. How do scientists actually know these ages? Geologic age dating—assigning an age to materials—is an entire discipline of its own. In a way this field, called geochronology, is some of the purest detective work earth scientists do. There are two basic approaches: relative age dating, and absolute age dating. Here is an easy-to understand analogy for your students: relative age dating is like saying that your grandfather is older than you. Absolute age dating is like saying you are 15 years old and your grandfather is 77 years old. To determine the relative age of different rocks, geologists start with the assumption that unless something has happened, in a sequence of sedimentary rock layers, the newer rock layers will be on top of older ones. This is called the Rule of Superposition. This rule is common sense, but it serves as a powerful reference point. Relative age dating also means paying attention to crosscutting relationships. Say for example that a volcanic dike, or a fault, cuts across several sedimentary layers, or maybe through another volcanic rock type. Pretty obvious that the dike came after the rocks it cuts through, right? With absolute age dating, you get a real age in actual years. First, the fossils. Based on the Rule of Superposition, certain organisms clearly lived before others, during certain geologic times. The narrower a range of time that an animal lived, the better it is as an index of a specific time. No bones about it, fossils are important age markers. But the most accurate forms of absolute age dating are radiometric methods. This rate of decay is called a half-life. So geochronolgists just measure the ratio of the remaining parent atom to the amount of daughter and voila, they know how long the molecule has been hanging out decaying. There are a couple catches, of course. Not all rocks have radioactive elements. Sedimentary rocks in particular are notoriously radioactive-free zones. So to date those, geologists look for layers like volcanic ash that might be sandwiched between the sedimentary layers, and that tend to have radioactive elements. Each radioactive isotope works best for particular applications. The half-life of carbon 14, for example, is 5,730 years. On the other hand, the half-life of the isotope potassium 40 as it decays to argon is 1. If a rock has been partially melted, or otherwise metamorphosed, that causes complications for radiometric absolute age dating as well. Have students reconstruct a simple geologic history — which are the oldest rocks shown? Which are the youngest? Take students on a neighborhood walk and see what you can observe about age dates around you. For example, which is older, the bricks in a building or the building itself? Are there repairs or cracks in the sidewalk that came after the sidewalk was built? Absolute age dating : Have students work alone or in pairs to find an article or paper that uses radiometric age dating. Which for youngest? Can you tell why? Secure Server - We value your privacy. Search Kids Discover. All Blog Posts. Quick View. Print Title. Free Download. Lesson Plan. Already a Member, Log In:. Register below:. Lost your password? Don't have an account? Sign up now, it's FREE. Log In Register Lost password.
Geochronology is the science of determining the age of rocks , fossils , and sediments using signatures inherent in the rocks themselves. Absolute geochronology can be accomplished through radioactive isotopes , whereas relative geochronology is provided by tools such as palaeomagnetism and stable isotope ratios. By combining multiple geochronological and biostratigraphic indicators the precision of the recovered age can be improved. Biostratigraphy does not directly provide an absolute age determination of a rock, but merely places it within an interval of time at which that fossil assemblage is known to have coexisted. Both disciplines work together hand in hand, however, to the point where they share the same system of naming strata rock layers and the time spans utilized to classify sublayers within a stratum. The science of geochronology is the prime tool used in the discipline of chronostratigraphy , which attempts to derive absolute age dates for all fossil assemblages and determine the geologic history of the Earth and extraterrestrial bodies. By measuring the amount of radioactive decay of a radioactive isotope with a known half-life , geologists can establish the absolute age of the parent material. A number of radioactive isotopes are used for this purpose, and depending on the rate of decay, are used for dating different geological periods. More slowly decaying isotopes are useful for longer periods of time, but less accurate in absolute years. A series of related techniques for determining the age at which a geomorphic surface was created exposure dating , or at which formerly surficial materials were buried burial dating. Exposure dating uses the concentration of exotic nuclides e. Many types of luminescence techniques are utilized in geology, including optically stimulated luminescence OSL , cathodoluminescence CL , and thermoluminescence TL. Thermoluminescence and optically stimulated luminescence are used in archaeology to date 'fired' objects such as pottery or cooking stones and can be used to observe sand migration. Incremental dating techniques allow the construction of year-by-year annual chronologies, which can be fixed i. A sequence of paleomagnetic poles usually called virtual geomagnetic poles , which are already well defined in age, constitutes an apparent polar wander path APWP. Such a path is constructed for a large continental block. APWPs for different continents can be used as a reference for newly obtained poles for the rocks with unknown age. Two methods of paleomagnetic dating have been suggested: 1 the angular method and 2 the rotation method. The second method is used for the folded areas where tectonic rotations are possible. The polarity timescale has been previously determined by dating of seafloor magnetic anomalies, radiometrically dating volcanic rocks within magnetostratigraphic sections, and astronomically dating magnetostratigraphic sections. Global trends in isotope compositions, particularly carbon-13 and strontium isotopes, can be used to correlate strata. Marker horizons are stratigraphic units of the same age and of such distinctive composition and appearance, that despite their presence in different geographic sites, there is certainty about their age-equivalence. Fossil faunal and floral assemblages , both marine and terrestrial, make for distinctive marker horizons. Tephra is also often used as a dating tool in archaeology, since the dates of some eruptions are well-established. It is important not to confuse geochronologic and chronostratigraphic units. Science of determining the age of rocks, sediments and fossils. Main article: Radiometric dating. Main article: Fission track dating. Main article: Cosmogenic radionuclide dating. Main article: Incremental dating. Main article: Magnetostratigraphy. Radiogenic Isotope Geology. Cambridge, Cambridge University Press. Principles of isotope geology. Earth and Planetary Science Letters. Evolutionary Anthropology. Science , 277 , 1279-1280 "Archived copy" PDF. Retrieved 2008-10-25. Tectonophysics, doi: 10. Canadian Journal of Earth Sciences. Doklady Earth Sciences. Outline of geology Glossary of geology History of geology Index of geology articles. Geochronology Geological history of Earth Timeline of geology. Geochemistry Crystallography Mineralogy Petrology Sedimentology. Stratigraphy Paleontology Paleoclimatology Palaeogeography. Structural geology Geodynamics Plate tectonics Geomorphology Volcanology. Glaciology Hydrogeology Marine geology. Geodesy Geomagnetism Geophysical survey Seismology Tectonophysics. Geologist Petroleum geologist Volcanologist. Geology Earth sciences Geology. Past history deep time Present Future Futures studies Far future in religion Timeline of the far future Eternity Eternity of the world. Horology History of timekeeping devices Main types astrarium atomic quantum hourglass marine sundial sundial markup schema watch mechanical stopwatch water-based Cuckoo clock Digital clock Grandfather clock. Chronology History. Religion Mythology. Geological time age chron eon epoch era period Geochronology Geological history of Earth. Chronological dating Chronobiology Circadian rhythms Dating methodologies in archaeology Time geography. Category Commons. Periods Eras Epochs. Canon of Kings Lists of kings Limmu. Chinese Japanese Korean Vietnamese. Lunisolar Solar Lunar Astronomical year numbering. Deep time Geological history of Earth Geological time units. Chronostratigraphy Geochronology Isotope geochemistry Law of superposition Luminescence dating Samarium—neodymium dating. Amino acid racemisation Archaeomagnetic dating Dendrochronology Ice core Incremental dating Lichenometry Paleomagnetism Radiometric dating Radiocarbon Uranium—lead Potassium—argon Tephrochronology Luminescence dating Thermoluminescence dating. Fluorine absorption Nitrogen dating Obsidian hydration Seriation Stratigraphy. Molecular clock. Categories : Geochronology Radiometric dating. Namespaces Article Talk. Views Read Edit View history. Help Learn to edit Community portal Recent changes Upload file. Download as PDF Printable version. Wikimedia Commons. Geology Geological time age chron eon epoch era period Geochronology Geological history of Earth. Concepts Deep time Geological history of Earth Geological time units. Absolute dating Amino acid racemisation Archaeomagnetic dating Dendrochronology Ice core Incremental dating Lichenometry Paleomagnetism Radiometric dating Radiocarbon Uranium—lead Potassium—argon Tephrochronology Luminescence dating Thermoluminescence dating.
Radiometric dating , radioactive dating or radioisotope dating is a technique which is used to date materials such as rocks or carbon , in which trace radioactive impurities were selectively incorporated when they were formed. Together with stratigraphic principles , radiometric dating methods are used in geochronology to establish the geologic time scale. By allowing the establishment of geological timescales, it provides a significant source of information about the ages of fossils and the deduced rates of evolutionary change. Radiometric dating is also used to date archaeological materials, including ancient artifacts. Different methods of radiometric dating vary in the timescale over which they are accurate and the materials to which they can be applied. All ordinary matter is made up of combinations of chemical elements , each with its own atomic number , indicating the number of protons in the atomic nucleus. Additionally, elements may exist in different isotopes , with each isotope of an element differing in the number of neutrons in the nucleus. A particular isotope of a particular element is called a nuclide. Some nuclides are inherently unstable. That is, at some point in time, an atom of such a nuclide will undergo radioactive decay and spontaneously transform into a different nuclide. This transformation may be accomplished in a number of different ways, including alpha decay emission of alpha particles and beta decay electron emission, positron emission, or electron capture. Another possibility is spontaneous fission into two or more nuclides. While the moment in time at which a particular nucleus decays is unpredictable, a collection of atoms of a radioactive nuclide decays exponentially at a rate described by a parameter known as the half-life , usually given in units of years when discussing dating techniques. In many cases, the daughter nuclide itself is radioactive, resulting in a decay chain , eventually ending with the formation of a stable nonradioactive daughter nuclide; each step in such a chain is characterized by a distinct half-life. In these cases, usually the half-life of interest in radiometric dating is the longest one in the chain, which is the rate-limiting factor in the ultimate transformation of the radioactive nuclide into its stable daughter. For most radioactive nuclides, the half-life depends solely on nuclear properties and is essentially constant. It is not affected by external factors such as temperature , pressure , chemical environment, or presence of a magnetic or electric field. This allows one to measure a very wide range of ages. Isotopes with very long half-lives are called "stable isotopes," and isotopes with very short half-lives are known as "extinct isotopes. The radioactive decay constant, the probability that an atom will decay per year, is the solid foundation of the common measurement of radioactivity. The accuracy and precision of the determination of an age and a nuclide's half-life depends on the accuracy and precision of the decay constant measurement. A faster method involves using particle counters to determine alpha, beta or gamma activity, and then dividing that by the number of radioactive nuclides. However, it is challenging and expensive to accurately determine the number of radioactive nuclides. Alternatively, decay constants can be determined by comparing isotope data for rocks of known age. This method requires at least one of the isotope systems to be very precisely calibrated, such as the Pb-Pb system. The possible confounding effects of contamination of parent and daughter isotopes have to be considered, as do the effects of any loss or gain of such isotopes since the sample was created. It is therefore essential to have as much information as possible about the material being dated and to check for possible signs of alteration. This can reduce the problem of contamination. In uranium—lead dating , the concordia diagram is used which also decreases the problem of nuclide loss. Finally, correlation between different isotopic dating methods may be required to confirm the age of a sample. The procedures used to isolate and analyze the parent and daughter nuclides must be precise and accurate. This normally involves isotope-ratio mass spectrometry. The precision of a dating method depends in part on the half-life of the radioactive isotope involved. For instance, carbon-14 has a half-life of 5,730 years. After an organism has been dead for 60,000 years, so little carbon-14 is left that accurate dating cannot be established. On the other hand, the concentration of carbon-14 falls off so steeply that the age of relatively young remains can be determined precisely to within a few decades. The closure temperature or blocking temperature represents the temperature below which the mineral is a closed system for the studied isotopes. If a material that selectively rejects the daughter nuclide is heated above this temperature, any daughter nuclides that have been accumulated over time will be lost through diffusion , resetting the isotopic "clock" to zero. As the mineral cools, the crystal structure begins to form and diffusion of isotopes is less easy. At a certain temperature, the crystal structure has formed sufficiently to prevent diffusion of isotopes. Thus an igneous or metamorphic rock or melt, which is slowly cooling, does not begin to exhibit measurable radioactive decay until it cools below the closure temperature. The age that can be calculated by radiometric dating is thus the time at which the rock or mineral cooled to closure temperature. These temperatures are experimentally determined in the lab by artificially resetting sample minerals using a high-temperature furnace. This field is known as thermochronology or thermochronometry. The mathematical expression that relates radioactive decay to geologic time is [14] [16]. The equation is most conveniently expressed in terms of the measured quantity N t rather than the constant initial value N o. The above equation makes use of information on the composition of parent and daughter isotopes at the time the material being tested cooled below its closure temperature. This is well-established for most isotopic systems. An isochron plot is used to solve the age equation graphically and calculate the age of the sample and the original composition. Radiometric dating has been carried out since 1905 when it was invented by Ernest Rutherford as a method by which one might determine the age of the Earth. In the century since then the techniques have been greatly improved and expanded. The mass spectrometer was invented in the 1940s and began to be used in radiometric dating in the 1950s. The ions then travel through a magnetic field, which diverts them into different sampling sensors, known as " Faraday cups ", depending on their mass and level of ionization. On impact in the cups, the ions set up a very weak current that can be measured to determine the rate of impacts and the relative concentrations of different atoms in the beams. Uranium—lead radiometric dating involves using uranium-235 or uranium-238 to date a substance's absolute age. This scheme has been refined to the point that the error margin in dates of rocks can be as low as less than two million years in two-and-a-half billion years. Uranium—lead dating is often performed on the mineral zircon ZrSiO 4 , though it can be used on other materials, such as baddeleyite , as well as monazite see: monazite geochronology. Zircon has a very high closure temperature, is resistant to mechanical weathering and is very chemically inert. Zircon also forms multiple crystal layers during metamorphic events, which each may record an isotopic age of the event. One of its great advantages is that any sample provides two clocks, one based on uranium-235's decay to lead-207 with a half-life of about 700 million years, and one based on uranium-238's decay to lead-206 with a half-life of about 4. This can be seen in the concordia diagram, where the samples plot along an errorchron straight line which intersects the concordia curve at the age of the sample. This involves the alpha decay of 147 Sm to 143 Nd with a half-life of 1. Accuracy levels of within twenty million years in ages of two-and-a-half billion years are achievable. This involves electron capture or positron decay of potassium-40 to argon-40. Potassium-40 has a half-life of 1. This is based on the beta decay of rubidium-87 to strontium-87 , with a half-life of 50 billion years. This scheme is used to date old igneous and metamorphic rocks , and has also been used to date lunar samples. Closure temperatures are so high that they are not a concern. Rubidium-strontium dating is not as precise as the uranium-lead method, with errors of 30 to 50 million years for a 3-billion-year-old sample. Application of in situ analysis Laser-Ablation ICP-MS within single mineral grains in faults have shown that the Rb-Sr method can be used to decipher episodes of fault movement. A relatively short-range dating technique is based on the decay of uranium-234 into thorium-230, a substance with a half-life of about 80,000 years. It is accompanied by a sister process, in which uranium-235 decays into protactinium-231, which has a half-life of 32,760 years. The scheme has a range of several hundred thousand years. A related method is ionium—thorium dating , which measures the ratio of ionium thorium-230 to thorium-232 in ocean sediment. Radiocarbon dating is also simply called carbon-14 dating. Carbon-14 is a radioactive isotope of carbon, with a half-life of 5,730 years [28] [29] which is very short compared with the above isotopes , and decays into nitrogen. Carbon-14, though, is continuously created through collisions of neutrons generated by cosmic rays with nitrogen in the upper atmosphere and thus remains at a near-constant level on Earth. The carbon-14 ends up as a trace component in atmospheric carbon dioxide CO 2. A carbon-based life form acquires carbon during its lifetime. When an organism dies, it ceases to take in new carbon-14, and the existing isotope decays with a characteristic half-life 5730 years. The proportion of carbon-14 left when the remains of the organism are examined provides an indication of the time elapsed since its death. This makes carbon-14 an ideal dating method to date the age of bones or the remains of an organism. The carbon-14 dating limit lies around 58,000 to 62,000 years. The rate of creation of carbon-14 appears to be roughly constant, as cross-checks of carbon-14 dating with other dating methods show it gives consistent results. However, local eruptions of volcanoes or other events that give off large amounts of carbon dioxide can reduce local concentrations of carbon-14 and give inaccurate dates. The releases of carbon dioxide into the biosphere as a consequence of industrialization have also depressed the proportion of carbon-14 by a few percent; conversely, the amount of carbon-14 was increased by above-ground nuclear bomb tests that were conducted into the early 1960s. Also, an increase in the solar wind or the Earth's magnetic field above the current value would depress the amount of carbon-14 created in the atmosphere. This involves inspection of a polished slice of a material to determine the density of "track" markings left in it by the spontaneous fission of uranium-238 impurities. The uranium content of the sample has to be known, but that can be determined by placing a plastic film over the polished slice of the material, and bombarding it with slow neutrons. This causes induced fission of 235 U, as opposed to the spontaneous fission of 238 U. The fission tracks produced by this process are recorded in the plastic film. This scheme has application over a wide range of geologic dates. Older materials can be dated using zircon , apatite , titanite , epidote and garnet which have a variable amount of uranium content. The technique has potential applications for detailing the thermal history of a deposit. The residence time of 36 Cl in the atmosphere is about 1 week. Thus, as an event marker of 1950s water in soil and ground water, 36 Cl is also useful for dating waters less than 50 years before the present. Luminescence dating methods are not radiometric dating methods in that they do not rely on abundances of isotopes to calculate age. Instead, they are a consequence of background radiation on certain minerals. Over time, ionizing radiation is absorbed by mineral grains in sediments and archaeological materials such as quartz and potassium feldspar. The radiation causes charge to remain within the grains in structurally unstable "electron traps". Exposure to sunlight or heat releases these charges, effectively "bleaching" the sample and resetting the clock to zero. The trapped charge accumulates over time at a rate determined by the amount of background radiation at the location where the sample was buried. Stimulating these mineral grains using either light optically stimulated luminescence or infrared stimulated luminescence dating or heat thermoluminescence dating causes a luminescence signal to be emitted as the stored unstable electron energy is released, the intensity of which varies depending on the amount of radiation absorbed during burial and specific properties of the mineral. Pottery shards can be dated to the last time they experienced significant heat, generally when they were fired in a kiln. Absolute radiometric dating requires a measurable fraction of parent nucleus to remain in the sample rock. For rocks dating back to the beginning of the solar system, this requires extremely long-lived parent isotopes, making measurement of such rocks' exact ages imprecise. At the beginning of the solar system, there were several relatively short-lived radionuclides like 26 Al, 60 Fe, 53 Mn, and 129 I present within the solar nebula. Dating methods based on extinct radionuclides can also be calibrated with the U-Pb method to give absolute ages. Thus both the approximate age and a high time resolution can be obtained. Generally a shorter half-life leads to a higher time resolution at the expense of timescale. The iodine-xenon chronometer [35] is an isochron technique. Samples are exposed to neutrons in a nuclear reactor. This converts the only stable isotope of iodine 127 I into 128 Xe via neutron capture followed by beta decay of 128 I. After irradiation, samples are heated in a series of steps and the xenon isotopic signature of the gas evolved in each step is analysed. This in turn corresponds to a difference in age of closure in the early solar system. Another example of short-lived extinct radionuclide dating is the 26 Al — 26 Mg chronometer, which can be used to estimate the relative ages of chondrules. The 26 Al — 26 Mg chronometer gives an estimate of the time period for formation of primitive meteorites of only a few million years 1. Technique used to date materials such as rocks or carbon. See also: Radioactive decay law. Main article: Closure temperature. Main article: Uranium—lead dating. Main article: Samarium—neodymium dating. Main article: Potassium—argon dating. Main article: Rubidium—strontium dating. Main article: Uranium—thorium dating. Main article: Radiocarbon dating. Main article: fission track dating. Main article: Luminescence dating. Earth sciences portal Geophysics portal Physics portal. Part II. American Journal of Science. Bibcode : 1907AmJS... In Roth, Etienne; Poty, Bernard eds. Nuclear Methods of Dating. Springer Netherlands. Applied Radiation and Isotopes. Annual Review of Nuclear Science. Bibcode : 1976Natur. January 2001. Geochimica et Cosmochimica Acta. Bibcode : 2001GeCoA.. Earth and Planetary Science Letters. Brent 1994. The age of the earth. Stanford, Calif. Radiogenic isotope geology 2nd ed. Cambridge: Cambridge Univ. Principles and applications of geochemistry: a comprehensive textbook for geology students 2nd ed. Using geochemical data: evaluation, presentation, interpretation. Harlow : Longman. Cornell University. United States Geological Survey. Kramers June 1975. Hanson; M. Martin; S. Bowring; H. Jelsma; P. Dirks 2001. Journal of African Earth Sciences. Bibcode : 2001JAfES.. Precambrian Research. Bibcode : 2002PreR.. Vetter; Donald W. Davis 2004. Bibcode : 2004JAfES.. Chemical Geology. Bibcode : 2001ChGeo. South African Journal of Geology. Wilson; R. Carlson December 1998. In situ Rb-Sr dating of slickenfibres in deep crystalline basement faults. Sci Rep 10, 562 2020. The Swedish National Heritage Board. Retrieved 9 March 2009. Dergachev 2002. Annales Geophysicae. Bibcode : 2002AnGeo.. Retrieved 6 April 2016. Thomas August 2001. Lissauer: Planetary Sciences , page 321. Cambridge University Press, 2001. V Pravdivtseva; A. Busfield; C. Hohenberg 2006. Meteoritics and Planetary Science. Lissauer: Planetary Sciences , page 322. Periods Eras Epochs. Canon of Kings Lists of kings Limmu. Chinese Japanese Korean Vietnamese. Lunisolar Solar Lunar Astronomical year numbering. Deep time Geological history of Earth Geological time units. Chronostratigraphy Geochronology Isotope geochemistry Law of superposition Luminescence dating Samarium—neodymium dating. Amino acid racemisation Archaeomagnetic dating Dendrochronology Ice core Incremental dating Lichenometry Paleomagnetism Radiometric dating Radiocarbon Uranium—lead Potassium—argon Tephrochronology Luminescence dating Thermoluminescence dating. Fluorine absorption Nitrogen dating Obsidian hydration Seriation Stratigraphy. Molecular clock. Categories : Radiometric dating Conservation and restoration of cultural heritage. Namespaces Article Talk. Views Read Edit View history. Help Learn to edit Community portal Recent changes Upload file. Download as PDF Printable version. Wikimedia Commons. Concepts Deep time Geological history of Earth Geological time units. Absolute dating Amino acid racemisation Archaeomagnetic dating Dendrochronology Ice core Incremental dating Lichenometry Paleomagnetism Radiometric dating Radiocarbon Uranium—lead Potassium—argon Tephrochronology Luminescence dating Thermoluminescence dating.